Methodology
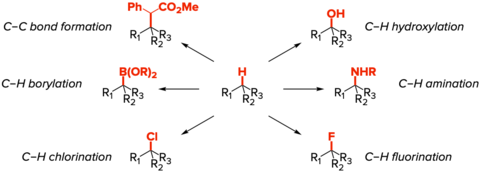
In the Du Bois lab, we are particularly interested in developing selective C–H functionalization processes for rapid generation of complex molecular architectures through site-specific modification of aliphatic C–H bonds. We have applied these selective processes and new catalyst systems to the synthesis of a number of natural products, including tetrodotoxin, manzacidins, and agelastatin. We are continually evolving the capabilities of these powerful reactions and evaluating their performance in total syntheses.
Despite the considerable advances in the capabilities of C-H functionalization over the past few years, numerous challenges remain to be solved if these technologies are to become part of the standard lexicon of chemical synthesis. In particular, the ability to control at the reagent-level with high precision the site of C–H bond modification in complex structures remains an unsolved problem.
We have investigated novel dirhodium and diruthenium complexes for C–H amination and both ruthenium and non-metal-based oxidants for C–H hydroxylation, and are actively involved in improving these systems as well as designing new ones. Detailed mechanistic investigations to identify factors that influence catalyst performance are central to this research. Efforts to utilize both amination and hydroxylation methods for the synthesis of molecules such as pactamycin and anisatin are on-going. These problems serve to demonstrate both at the strategic and tactical level the manner in which C–H functionalization can be employed to assemble molecules.
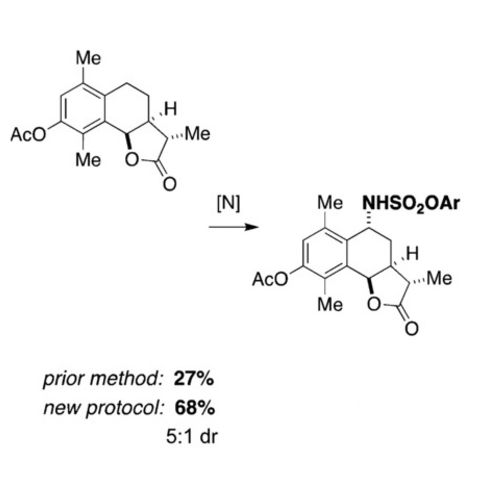
C-H Amination
The Du Bois lab has a long-standing interest in catalytic C–H amination using metal nitrenoids. Through many years of catalyst development and mechanistic investigations, we have developed systems capable of functionalizing complex molecules through both intra- and intermolecular means. Our efforts have led to a number of different ligand designs for the dirhodium paddlewheel complexes, including carboxylates and amidates, among others, with corresponding advances in the reaction technology. New catalyst design is an ongoing interest in the Du Bois lab and we seek to create new catalysts capable of new selectivities and differential reaction performance to complement currently-established methods.
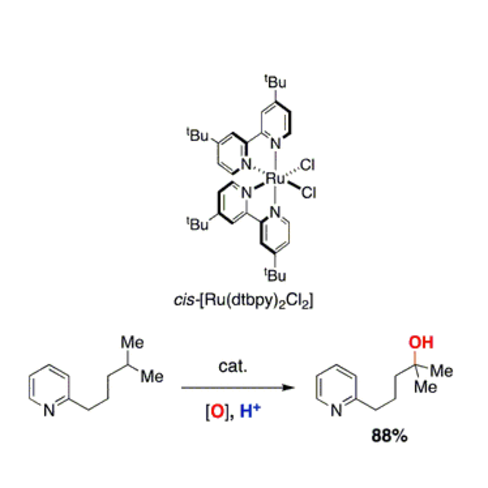
C-H Hydroxylation
We are interested in developing ruthenium catalysts for C–H hydroxylation. Recently, we developed a system capable of oxidizing C–H bonds in the presence of basic amines using in situ protonation and a water-soluble catalyst. We are actively interested in improving this system and developing hydroxylation catalysts capable of functionalizing other diverse substrates. Mechanistic investigations have been key in developing this reaction technology.